Highlighted articles
Hot off the press
Peti & Page (2013) Molecular Basis of MAPK Regulation Protein Science, in press.
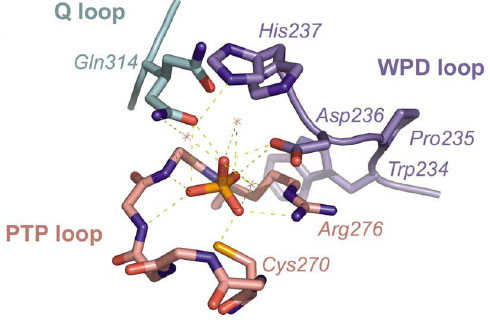
Peti & Page (2013): Enzyme Mechanisms: What's up Doc?, Nature Chemical Biol, in press.
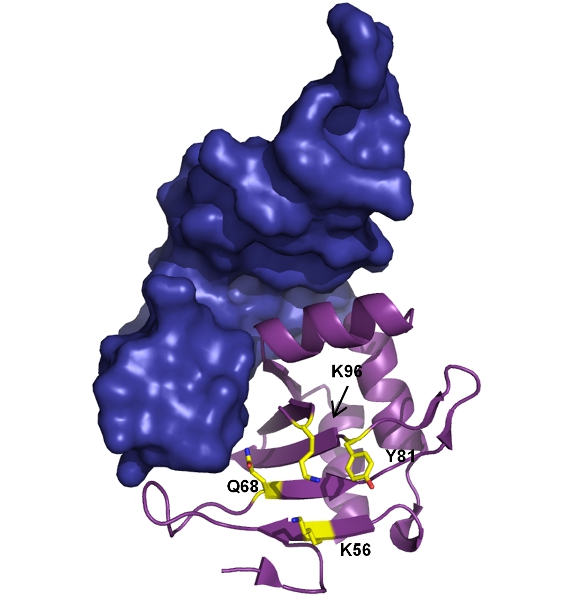
Francis, DM et al. (2013) The differential regulation of p38 by the neuronal kinase interaction motif PTPs, a detailed molecular studyStructure, 9:1612-1623.
Selected Articles (of 88)
Wang, Lord et al. (2012) A new type V toxin-antitoxin system where mRNA for toxin GhoT is cleaved by antitoxin GhoS. Nature Chem Biol 8:855-861.
Grigoriu, et al. (2013) The molecular mechanism of substrate engagement and immunosuppresent inhibitionof calcineurin. Plos Biol. 11: e1001492.
Francis, DM, Rozycki, B, Koveal, D, Hummer, G, Page, R, Peti, W (2011) Structural basis of p38α regulation by hematopoietic tyrosine phosphatase. Nature Chem Biol 7:916-924.
Ragusa, MJ et al. (2010). Spinophilin directs protein phosphatase 1 specificity by blocking substrate binding sites. Nature Struct Mol Biol 17:459-464.
Brown, BL, Grigoriu, S, Kim, Y, Arruda, JM, Davenport, A, Wood, TK, Peti, W, Page, R (2009) Three dimensional structure of the MqsR:MqsA complex: a novel TA pair comprised of a toxin homologous to RelE and an antitoxin with unique properties. PLoS Pathogens 5:12. e1000706.
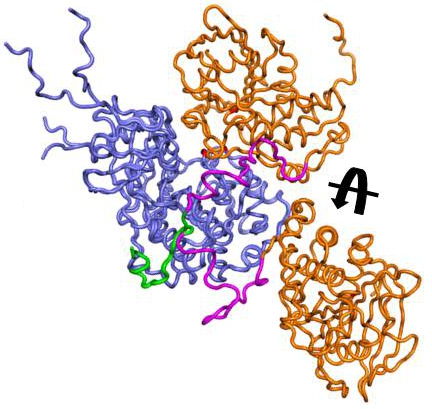
The MAP kinase ERK2 is regulated by numerous phosphatases that tightly control its activity. For example, the hematopoietic tyrosine phosphatase (HePTP) negatively regulates T cell activation in lymphocytes via ERK2 regulation. However, only very limited structural information is available for these biologically important complexes. Here, we use small angle X-ray scattering combined with EROS ensemble refinement to characterize the resting and active states of HePTP:ERK2 complex structures. Our data shows that the resting state HePTP:ERK2 complex adopts a highly extended, dynamic conformation that becomes compact and ordered in the active state complex. This work experimentally demonstrates that these complexes undergo significant dynamic structural changes in solution and provides the first structural insight into an active state MAPK complex.
From: Francis, D.M., Rozycki, B., Tortajada, A., Hummer, G., Peti, W., Page, R. (2011) Resting and Active States of the ERK2:HePTP Complex. JACS 133: 17138 – 17141.